In situ hydrogen production from hydrocarbon reservoirs – modelling study
因为氢气相比于碳氢化合物在环境方面具有许多好处,所以现在越来越多的人开始重视使用氢气,而不仅仅是考虑生产氢气所需的能源。尤其是在很多国家都设定了净零排放的目标的情况下,找到经济实惠、低碳的生产氢气的方法变得更加重要。来自卡尔加里大学的研究人员提出了一种创新的方法,可以从碳氢化合物储层中原位制氢,并同时将产生的副产品留在储层中。他们在研究中开发了一个考虑了四个关键的氢气生成反应的燃烧模型设计框架,包括蒸汽重整、部分氧化、自热重整和热解。研究人员使用化学平衡分析得到了一组非线性方程,并通过牛顿-雅可比迭代法求解这些方程。然后,他们通过分析每个反应的自由能变化,将其作为筛选模型的指标。燃烧模型的结果随后与使用热采模拟器CMG STARS得到的结果进行了验证。模型的结果表明,在原位氢气生产研究中,蒸汽碳比与产生的氢气量之间存在正弦关系。这个燃烧模型可以作为实验设计和分析的框架使用。
英国米德尔斯堡蒂赛德大学计算工程与数字技术学院
Abstract
The use of hydrogen is gaining reputation because of its many beneficial properties to the environment in comparison to hydrocarbon not minding its net energy requirement for production challenges. With most countries adopting a strategy to achieve their net-zero emissions targets, it becomes much more important to find affordable, low-carbon ways of producing hydrogen. An innovative method of producing hydrogen from hydrocarbon reservoirs while keeping the associated by-products in the reservoir has been demonstrated researchers from the University of Calgary. However, in this study, a framework for designing an in situ combustion model that considers four key hydrogen forming reactions – steam reforming, partial oxidation, autothermal reforming and pyrolysis, was developed. A set of non-linear equations obtained from chemical equilibrium analysis of the hydrogen forming reactions were solved using a Newton-Jacobi interation. Analysis of the change in Gibbs free energy of each reaction were then used as a screening tool for implementing a numerical model. Results obtained from the combustion model were then validated against results obtained from thermal reservoir simulator CMG STARS. Results from the model reveal an upward trending sinusoidal relationship between steam-carbon ratio and the amount of hydrogen yield from an in situ hydrogen production study. The combustion model could be used as a framework to design experimental analysis.

7 Conclusion
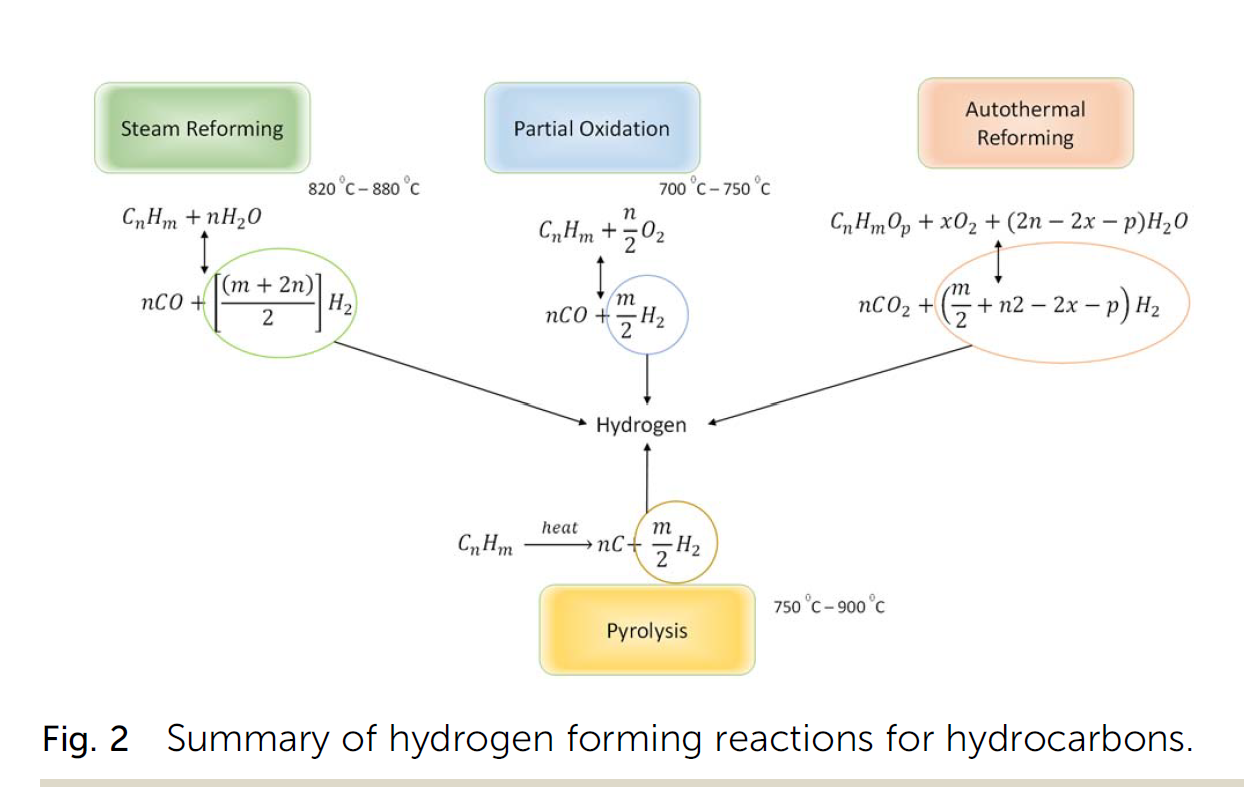
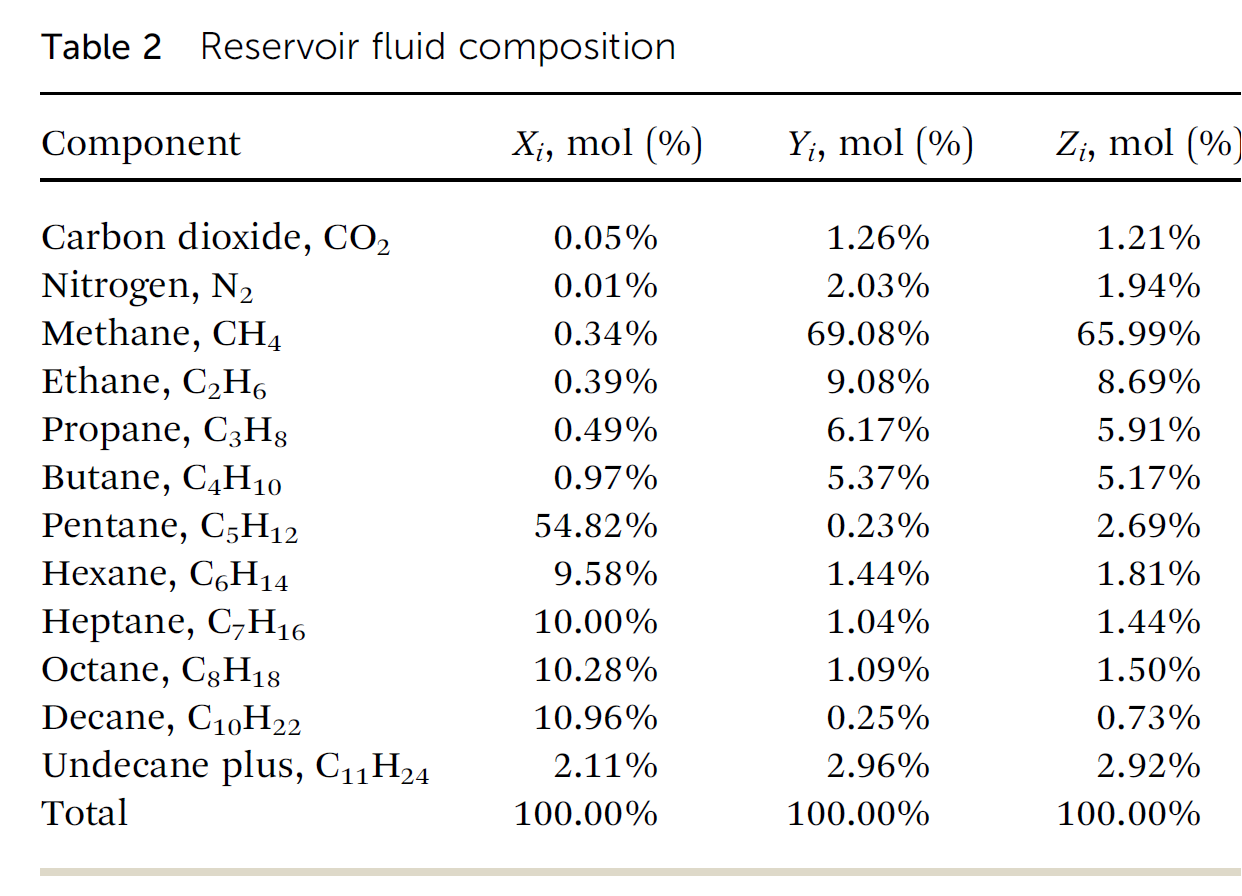
In situ hydrogen production (IHP) from hydrocarbon reservoirs is an interesting as well as exciting frontier for research. Applications of this novel process could be extended to existing oil/gas reservoirs and production facilities could be retrofitted to allow for hydrogen production. From literature, four broad types of hydrogen forming reactions occur in the reservoir – steam reforming, partial oxidation, autothermal reforming and pyrolysis. In this paper, a model for characterizing these hydrogen forming reactions under reservoir conditions was formulated based on composition of the reservoir fluid using equilibrium reaction analysis. Change in Gibbs free energy of each reaction, formed the basis of screening and implementation in a numerical simulator (CMG STARS). The results from the model were then validated against that obtained from the numerical simulator. Key findings from this study include:
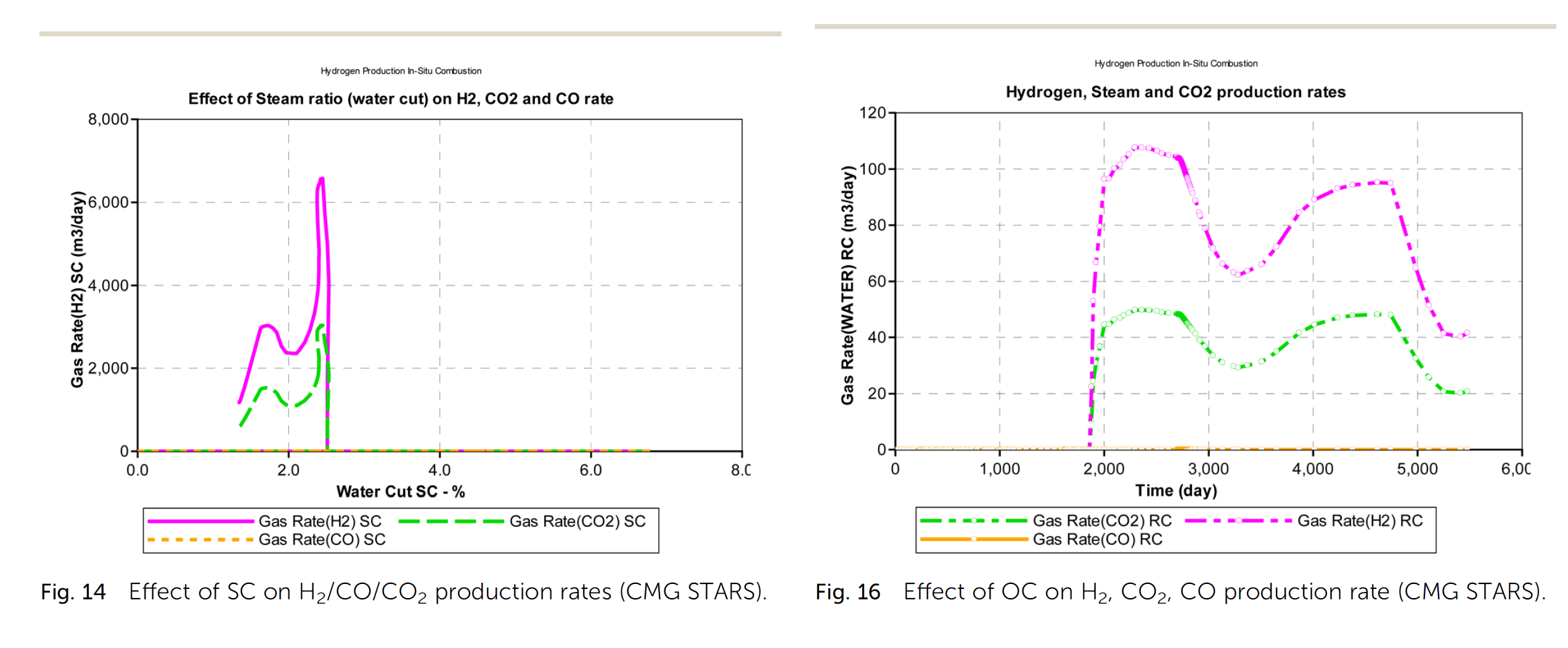
(i) The hydrogen yield from an in situ combustion process increases positively with the steam-carbon ratio during combustion. However, rather than a near exponential relationship between both parameters (hydrogen yield and steam-carbon ratio) as observed in most surface reactors, an upward trending sinusoidal relationship is observed.
(ii) In addition to the steam-carbon ratio, the oxygen–carbon ratio also affects the hydrogen yield from an in situ combustion process. A downward trending sinusoidal relation between the oxygen–carbon ratio and hydrogen yield is observed. Excess oxygen oxidates the produced hydrogen into steam.
The model developed in this paper sets the framework for future study in this direction and more importantly can be used to optimize preliminary experimental investigations. Future improvements in the combustion model could consider the effect of reservoir geology & rock mineralogy and temperature variations during combustion.
